Simulating tCS Electric Fields in the Brain
Contents
Introduction: the electric fields in the brain under tCS
We discuss here first the methodology for computing electric fields in the brain as implemented in our software. We then provide and overview as well as tips on use of our StimWeaver software and service.
For more information about the basic mechanisms and technologies in tCS, please see Ruffini et al. 2013 <ref> Ruffini, G., Wendling, F., Merlet, I., Molaee-Ardekani, B., Mekkonen, A., Salvador, R., Soria-Frisch, A., Grau, C., Dunne, S., Miranda, P., 2013. Transcranial current brain stimulation (tCS):models and technologies. IEEE Transactions on Neural Systems and Rehabilitation Engineering 21, 333–345.</ref> as well as this hive-eu.org project deliverable] <ref> P. C. Miranda (FFCUL), F. Wendling, I. Merlet, B. Molaee- Ardekani (INSERM), G. Ruffini (Ed.), S. Dunne, A. Soria- Frisch and D. Whitmer (Starlab), D1.1– Review of state-of-the-art in currents distri- bution and effects, HIVE FP7-FET Open project, 2009 </ref>.
A forthcoming paper will discuss the optimization of tCS montages from well defined cortical target maps <ref> Giulio Ruffini, Michael D. Fox, Oscar Ripolles, Pedro Cavaleiro Miranda, Alvaro Pascual-Leone, Optimization of multifocal transcranial current stimulation for weighted cortical pattern targeting from realistic modeling of electric fields, accepted for publication in Neuroimage 2014 </ref>
The mechanisms underlying the after-effects of tDCS are still the subject of investigation, but in all cases these local changes are brought about by the accumulated action of the applied electric field over time, directly or indirectly. For this reason we focus here on electric field optimization.
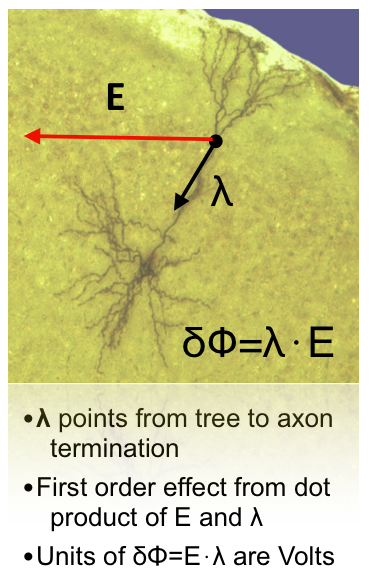
Moreover, given that that there are strong directional effects in the interaction of electric fields and neurons, i.e., neurons are influenced mostly by the component of the electric field parallel to their trajectory
<ref>Ranck, J., 1975. Which elements are excited in electrical stimulation of the mammalian central nervous system: a review. Brain Res 98, 417–440.</ref>
<ref>Rattay, F., 1986. Analysis of models for external stimulation of axons. IEEE Transactions on Biomedical Engineering 33, 974–977.</ref>
<ref> Rushton, W.A.H., 1927. The effect upon the threshold for nervous excitation of the length of nerve exposed, and the angle between current and nerve. J Physiol 63, 357–77.</ref>
<ref> Roth, B.J., 1994. Mechanisms for electrical stimulation of excitable tissue. Crit Rev Biomed Eng 22, 253–305.</ref>
<ref>Bikson, M., Inoue, M., Akiyama, H., Deans, J.K., Fox, J.E., Miyakawa, H., Jefferys, J.G., 2004. Effects of uniform extracellular dc electric fields on excitability in rat hippocampal slices in vitro. J Physiol 557, 175–90. </ref>
<ref> Fröhlich, F., McCormick, D.A., 2010. Endogenous electric fields may guide neocortical network activity. Neuron 67, 129–143.</ref>,
and that the effects of tDCS depend on its polarity, knowledge about the orientation of the electric field is crucial in predicting the effects of stimulation. The components of the field perpendicular and parallel to the cortical surface are of special importance, since pyramidal cells are mostly aligned perpendicular to the surface, while many cortical interneurons and axonal projections of pyramidal cells tend to align tangentially
<ref> Day, B., Dressler, D., Maertens de Noordhout, A., Marsden, C., Nakashima, K., Rothwell, J., Thompson, P., 1989. Electric and magnetic stimulation of human motor cortex: surface emg and single motor unit responses. J. Physiol 122, 449–473.</ref>
<ref> Fox, P.T., Narayana, S., Tandon, N., Sandoval, H., Fox, S.P., Kochunov, P., Lancaster, J.L., 2004. Column-based model of electric field excitation of cerebral cortex. Hum Brain Mapp 22, 1–14.</ref>
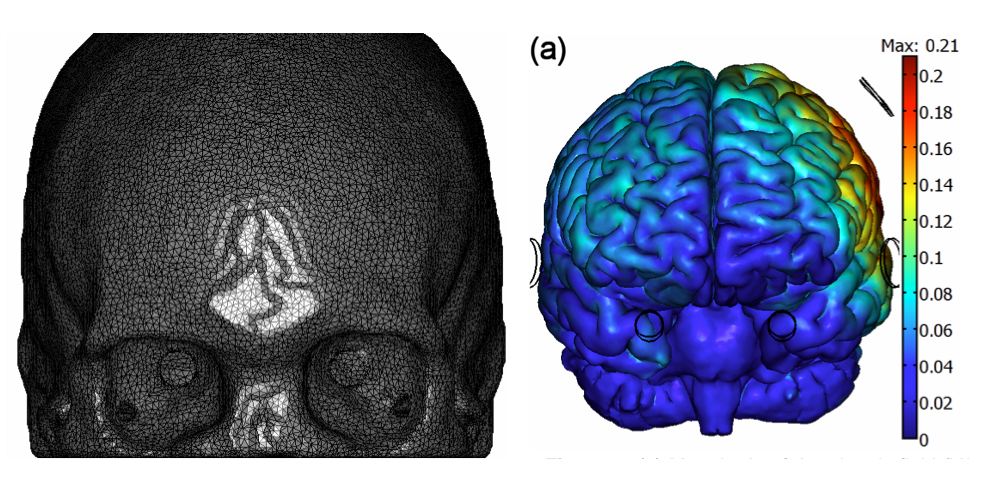
<ref> Kammer, T., Vorwerg, M., Herrnberger, B., 2007. Anisotropy in the visual cortex investigated by neuronavigated transcranial magnetic stimulation. Neuroimage 36, 313–321.</ref>. Thus, an important element in modeling is to provide the electric field distribution and orientation relative to the grey matter (GM) and white matter (WM) surfaces (the latter might be important to study the possibility of polarizing corticospinal axons, their collaterals and other projection neurons). In order to do this, we work here with a realistic head model derived from structural MRI images | Miranda et al. 2013 <ref name="Miranda2013a"> Miranda, P.C., Mekonnen, A., Salvador, R., Ruffini, G., 2013. The electric field in the cortex during transcranial current stimulation. Neuroimage 70, 45–58. </ref> to calculate the tCS electric field components rapidly from arbitrary EEG 10-20 montages. Importantly, this modeling approach allows for fast calculation of electric field components normal and parallel to the GM and WM surfaces.
StimViewer
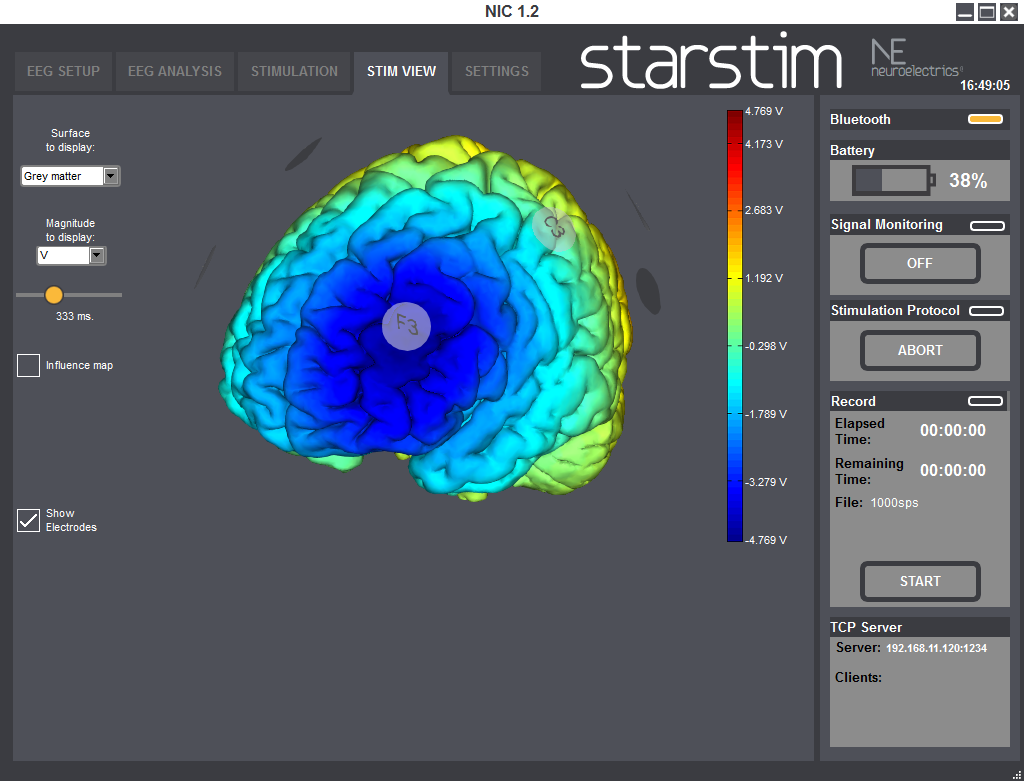
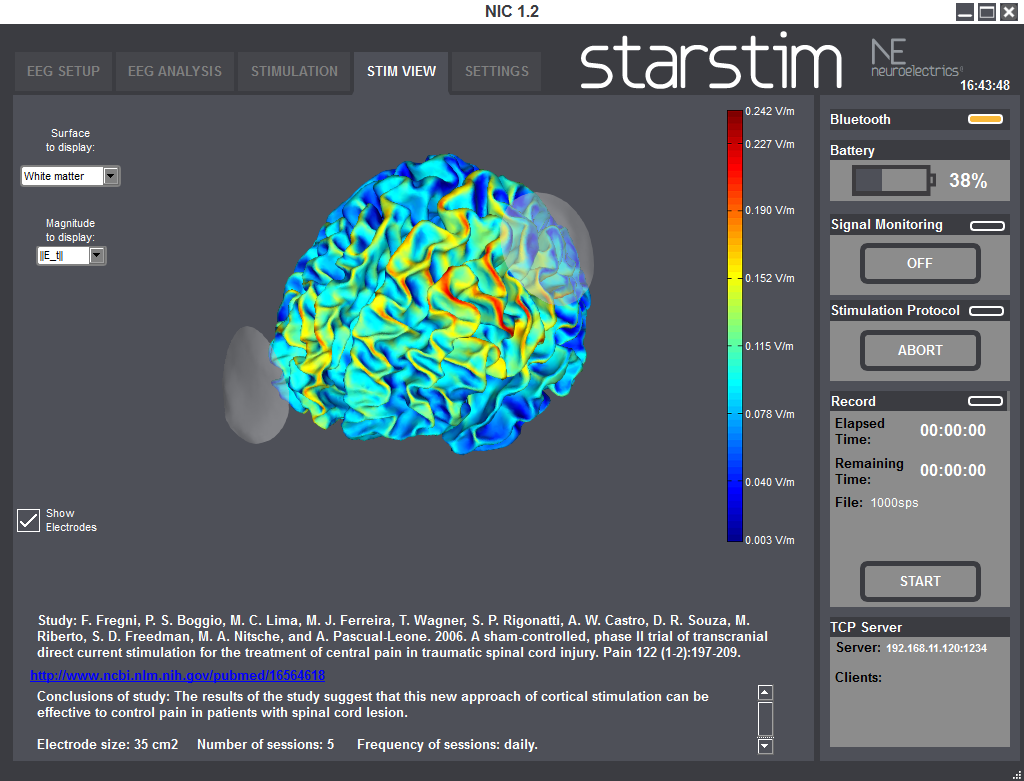
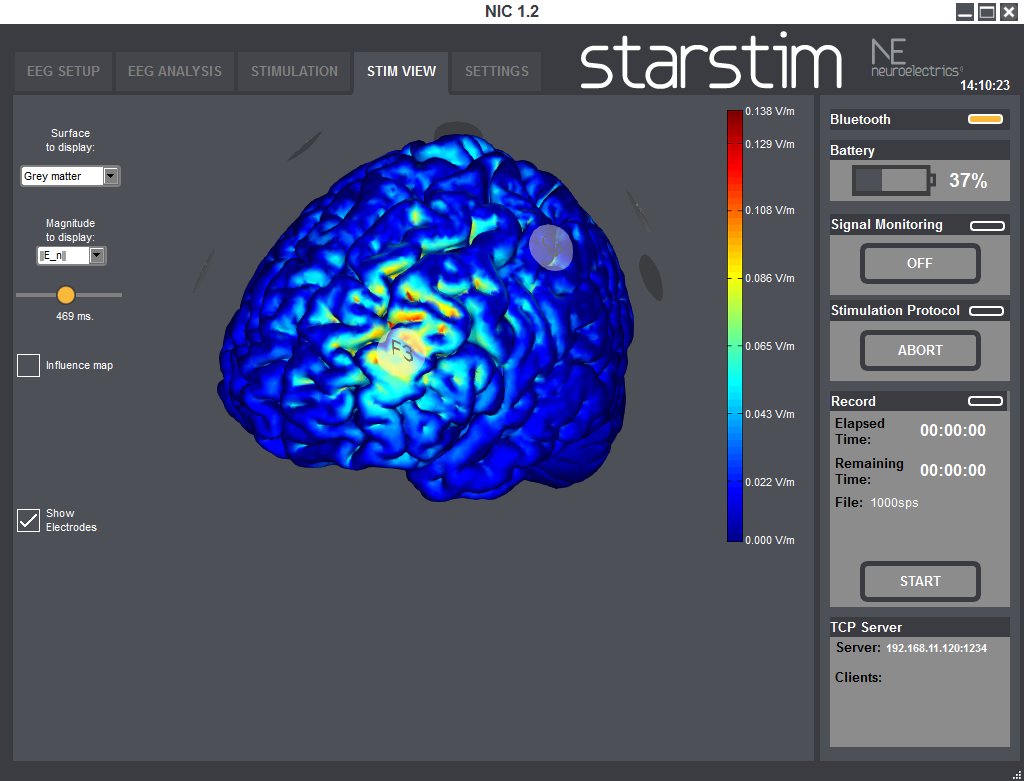
StimViewer is the software component embedded in | NIC which is activated when in use with | StarStim. StimViewer is a fast simulation engine to produce electric fields in the brain associated with a particular tCS montage and display them on two surfaces: the outer cortex or inner cortex. That results from the stimulation montage of each step of a stimulation protocol, is displayed in a 3D model.
The electric field calculations were performed using the realistic head model described in | Miranda et al. 2013aa <ref name="Miranda2013a" /> . Briefly, tissue boundaries were derived from MR images (scalp, skull, cerebrospinal fluid (CSF) – including ventricles, Grey Matter and White Matter) and the Finite Element Method was used to calculate the electric potential in the head, subject to the appropriate boundary conditions. Tissues were assumed to be uniform and isotropic and values for their electric conductivity were taken from the literature.
In order to compute electric fields rapidly, we have made use of the principle of superposition. This states that with appropriate boundary conditions, the solution to a general N-electrode problem can be expressed as a linear combination of N-1 bipolar ones. A fixed reference electrode is first chosen, and then all the bipolar solutions using this electrode are computed. A general solution with an arbitrary number of N electrodes can then easily be computed.
Using StimViewer you can display the electric Potential V and the normal (i.e., perpendicular) component of the electric field to the cortical surface (E_n, which can be positive or negative). In the convention used here, a positive value for the component of the electric field normal to the cortical surface means the electric field component normal is pointing into the cortex. According to the literature, such a field would be excitatory. On the other hand, an electric field pointing out of the cortex (negative normal component) would be inhibitory.
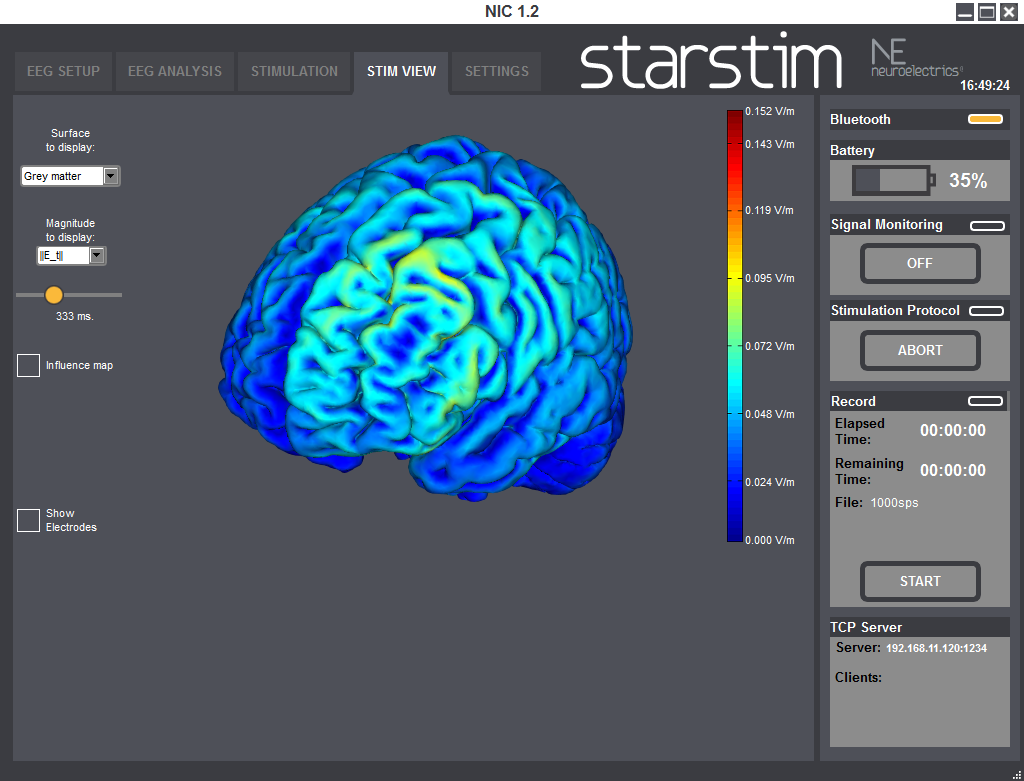
You can also display the magnitude of the tangential component (||E_t||), or the total magnitude (||E||).
For time varying protocols (such as tACS), you can also use a temporal slider to display the fields at different time points. Alternatively you can display the time average of the magnitude of the field, which we call the influence map.
StimWeaver
StimWeaver is an optimization service created by Neuroelectrics and based on StimViewer technology. It has been designed to create stimulation montages (electrode positions as well as currents) specifically adapted to user-defined targets.
Our staff will produce a mathematically optimized montage based on your specifications (target map, maximal currents, number of electrodes, etc.). Our optimization software (StimWeaver) carries out a thorough search in the space of electrode locations and currents to produce the best solution for the specified target.
Please see our recent paper on the subject of optimization of montages starting from well-defined cortical target maps <ref> Giulio Ruffini, Michael D. Fox, Oscar Ripolles, Pedro Cavaleiro Miranda, Alvaro Pascual-Leone, Optimization of multifocal transcranial current stimulation for weighted cortical pattern targeting from realistic modeling of electric fields, available online, Dec 15, Neuroimage (2013) </ref>.
Contact us at info@neuroelectrics.com for more information.
References
<references/>
References
<references />