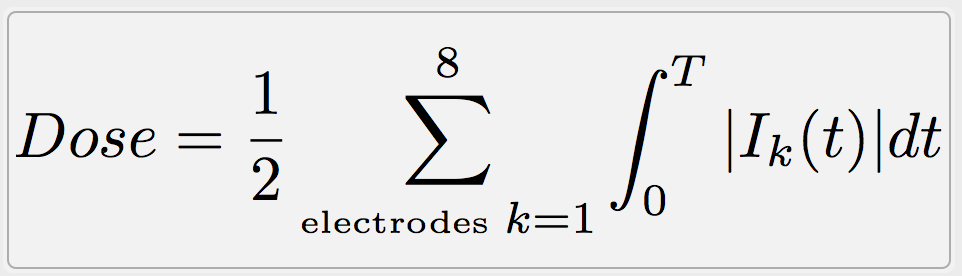Difference between revisions of "Safety"
(→StarStim) |
(→StarStim) |
||
| Line 50: | Line 50: | ||
[http://www.ncbi.nlm.nih.gov/pubmed/23123281 Faria P1, Fregni F, Sebastião F, Dias AI, Leal A., Feasibility of focal transcranial DC polarization with simultaneous EEG recording: preliminary assessment in healthy subjects and human epilepsy, Epilepsy Behav. 2012 Nov;25(3):417-25] | [http://www.ncbi.nlm.nih.gov/pubmed/23123281 Faria P1, Fregni F, Sebastião F, Dias AI, Leal A., Feasibility of focal transcranial DC polarization with simultaneous EEG recording: preliminary assessment in healthy subjects and human epilepsy, Epilepsy Behav. 2012 Nov;25(3):417-25] | ||
</ref>). | </ref>). | ||
| + | |||
Revision as of 07:15, 22 April 2015
Enobio
There are no special safety considerations for Enobio. However, for safety reasons, neither | Enobio nor | StarStim will operate while charging. In order to charge your NECBOX device, plug it via the USB port to a computer or power supply, and make sure the power switch of the NECBOX is in OFF position. If it is in the ON position, the device will not charge.
StarStim
Please note: tCS therapy should be supervised by a trained medical doctor. Modalities other than tDCS (tACS, tRNS) are for research purposes only.
Starstim is a CE medically certified product. However, it is not FDA approved for clinical use (tDCS, tACS or tRNS aren't approved as of yet) - it is classified as an investigational device under US federal law.
Built-in safety measures:
Maximal current at an electrode and voltage accross two points is limied (2 mA and 30 V respectively) Maximal injected current by all electrodes at any given time limited to 4 mA Continuous impedance check and limited application time (1h)
There are no studies in the literature describing the effects of direct current treatments on pregnant women, or children below 18 years, or patients with pacemakers, intracranial electrodes, implanted defibrillators, or any other prosthesis. Before applying | StarStim, make sure that pacemakers, intracranial electrodes, defibrillators, or any other prosthesis are not implanted in the patient. Otherwise, the application of DC currents could be unsafe.
For safety, Starstim is limited in the following ways. It will not operate if the contact impedance is above 20 kOhm. It can only provide potential differences across electrode of 30 V to deliver a maximum, at any electrode of (+ or -) 2 mA of current.
Our software allows for configurable, long, ramp up and ramp down times to maximize user comfort.
Based on abundant literature, the guideline for clinical use is to keep average current densities in electrodes below 2 mA/35 cm2= 0.06 mA/cm2. Such stimulation current densities are far from the threshold for tissue damage (14.3 mA/cm2) recently indicated for tDCS in an animal model. Typical applications times are of 20 min or less.
Current densities above 0.06 mA/cm2 (but always well below 14.3 mA/cm^2) are for advanced clinical or research purposes only.
Stimulation session durations beyond 40 minutes are for research purposes only.
In addition, electrode positions above cranial foramina and fissures should be avoided because these could increase beyond safety limits the effective current density.
| StarStim can only be used with specifically designed Neuroelectrics electrodes.
With sponge electrodes, the use of sodium chloride solution, the regular replacement of the sponge, and the careful inspection of the condition of the skin under the electrode before and after tDCS is recommended. Observed Adverse Effects include: skin itching, tingling, headache, burning sensation and discomfort. In rare cases, skin lesions have been observed. If skin lesions are observed, the treatment must be suspended and the equipment revised.
| StarStim components must never be opened or damaged. Before using check that | StarStim components, including electrodes, are undamaged and clean.
Safety Guide
The following chart is provided to guide operators, providing safety zones for different electrode current densities. Note this is an updated chart from the 2012 version. The only change is the removal of the "Uncharted Territory" zone. During the last year several groups have been working with our Pi electrodes using up to 2 mA of currents, and no ill effects have been reported.
It illustrates the MAX average current density that can be used for mainstream clinical applications, advanced clinical applications, research as a function of electrode size.
Please note: the proposed limits are not based on available negative evidence (i.e., findings of Adverse Effects with higher current densities). Rather, it is a conservative statement based on the limited experience with current densities above 2 mA/35 cm2).
With regards to the use of small electrodes in tCS, it is worthwhile noting that the ratio of current/electrode area (I/A ratio) is not a good indicator of cortical electric field magnitude. More specifically, in Miranda et al<ref> Pedro Cavaleiro Miranda, Paula Faria and Mark Hallett, What does the ratio of injected current to electrode area tell us about current density in the brain during tDCS?, Clin Neurophysiol. Jun 2009; 120(6): 1183–1187. </ref>, is is shown that for smaller electrodes, more current than predicted by the I/A ratio was required to achieve a predetermined current density in the brain.
In the last few years several studies have employed small electrodes with no ill side effects (e.g., Faria et al<ref>
Faria P1, Fregni F, Sebastião F, Dias AI, Leal A., Feasibility of focal transcranial DC polarization with simultaneous EEG recording: preliminary assessment in healthy subjects and human epilepsy, Epilepsy Behav. 2012 Nov;25(3):417-25
</ref>).
Dosage
For the stimulation using starstim, NIC gives an informative value about the dosage of a session. The dosage is the amount of charge that the user receives during the session. This calculation is done following the equation:
References
<references/>