It’s a dire truth that most drugs people are prescribed will fail to help them. Take the US top-selling medicament for high-cholesterol (Rosuvastatin), or the one routinely used to treat asthma (Fluticasone propionate): they can improve the conditions only for 1 patient in 20. And they are not exceptions. In fact, the success rate of the ten highest-grossing drugs in US ranges between 3 and 25%.1
The guidelines behind the use of these medications are derived from randomized clinical trials that must involve large and heterogeneous populations. If on one side these two requirements ensure the statistical relevance of the trial, on the other they miss or conceal the individual characteristics of the patient (genetics, environment and lifestyle) that might influence his/her specific response to the treatment.
This is why many physicians todays advocate for “precision medicine”, i.e. for therapies that take the individual variability into account 2.
Right, but what does this have to do with tCS?
Well, all considered, tCS is no different than a pill. In a pill, it’s active principle and the posology that make it effective. In tCS it’s the protocol of stimulation, or montage. The position of the electrodes and the current delivered create a flow of ion charges inside the head, and consequently a pattern of electric field (Efield), that modulates the neuronal response.
The optimal montage for a subject is the one producing the most effective stimulation on a pre-defined target area on his/her cortex.
So, let’s suppose you want to use tCS to stimulate a certain region of your cortex. You smartly choose to buy our NEStimweaver device, and…now what? What’s the montage you should use?
Are there guidelines for stimulation protocols that you can reference to? And how much should you rely on them?
And if your spouse or grandma or your overweight neighbor wanted to use the cap for the same purpose, would the same montage fit them?
In this blogpost, we’ll try to address these questions. BTW, please, do not just try it on your grandma. Keep on reading.
The optimal montage.
For simple targets, the problem of where to place the electrodes might appear quite straightforward and indeed for long tCS experts have placed just two big electrodes heuristically3 . However, calculations and recent in-vivo recordings have shown that the stimulation does not just occur under the active electrodes, as one would expect, but it is widely distributed and can include deep structures4. As a matter of fact, bipolar montages are seldomly the most effective in terms of focusing5, 6. Plus, when targeting complex and extended neural networks, multichannel montages are needed, whose configuration is far more difficult to determine by insight7.
To properly determine the position and the current at each electrode you must resort to mathematical algorithms of optimization. Various approaches can be followed, nicely summarized in Miranda’s review8 but the underlying principle is the same: minimizing the difference between the desired, ideal, Efield in the target region and the Efield created by the best achievable montage.
Standard montages
Today, it’s possible to find at least a validated montage for most target areas in the large heap of scientific publications on tCS9,10,11.
You might then be tempted to just adopt one such montages, assuming that replicating the stimulation parameters would ensure a consistent treatment outcome. Beware! Same as for many drugs, for tCS, this is not a minor assumption. Even when using the same stimulation protocol, tCS effects are extremely variable across individuals, to the point that its reliability as clinical tools keeps being questioned12. In the words of Wurtzman “the effect of tCS on any one individual is unpredictable. Up to 30% of subjects respond with changes in cortical excitability in the opposite direction from others, despite using identical setting”13.
Feeling appalled? Keep on reading.

Inter-subject variability
There are several reasons behind this inter-subject variability. As it often happens, the devil is in the details.
Firstly, the unique anatomy and tissue composition of everyone’s head. These have such a dramatic influence on the path followed by the currents, and therefore on the Efield distribution inside the brain, that the same montage might induce, in the same area, Efield up to three times different across subjects14,15.
The major actors seem to be head dimensions, gyrification morphology, sulcal depth and thickness of CSF and skull. The amount of fat in the scalp and of spongious bone in the skull, decreasing with age, also play a role, by affecting the conductivity of these tissues surrounding the brain8 (think of your grandma and neighbor).
Then, of course, unusual Efield distributions are to be expected in presence of neurological damages such as strokes, glioblastoma or craniotomies, and surgical implants. These necessarily alter the “normal” current pathways, in peculiar ways according to their distinctive nature.
And, things are even more complex: individuals can respond differently to the same montage even with consistent changes in cortical stimulation, i.e. even if the Efield on the target is similar16. Indeed, many other factors play a role: age, gender and hormones levels (mind your spouse), handedness, neurotransmitter levels, integrity and strength of network connections, fatigue, wakefulness, and, in case of lesions, etiology and time since injury13.
Personalized montages
When going for a montage to target a specific area, the more variables influencing the success of the stimulation you manage to control, the most reliable the outcome will be.
I guess at this point, you might feel overwhelmed by the task, but don’t despair. The good news is, that’s already possible, for the biophysical features.
In the last blogpost we explained how we can produce individual head models from MRI-T1 image segmentation. These models account for the individual head morphology, conductivity, and possible lesions or implants. They are used to calculate the current flow and Efield for a given stimulation.
When used in combination with montage optimizations algorithms, these models help us finding the most suitable configuration for the specific subject. We call this “personalized montage optimization”.
From this approach, we gain not only more stimulation focality on each individual of a population, but we also homogenize it across all of them. In other words, we make the treatment more reproducible.
In fact, controlling for the variability associated to the anatomy it’s essential to then go deeper, investigating all the other biological causes of variable tCS effects. By making this possible, personalized montages move us towards a clear assessment of the therapeutic value of tCS.
So, let’s go back to your initial questions. We’ve seen that merely applying a standard montage by the book, or even a montage optimized for someone else, is not enough to ensure tCS efficacy. And that a cleverer strategy is already available. Personalized optimization makes the most of realistic bespoke head models and produces montages that will maximize the chance of success for your treatment.
Click here to request one!

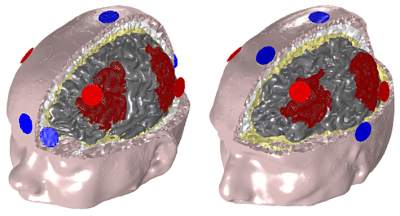
| Figure 1 The default montage exciting the frontoparietal network (in red) is applied to a MCS subject (left), and compared with the personalized optimization montage for the same subject and target area (right, in collaboration with www.luminous-project.eu). The personalized montage doubles the excitation on the target. Also, in a population of three subjects, the relative variability of the stimulation drops 50% for the personalized montages (not shown). |
| Figure 2 The personalized optimization montage takes into account the thickness of the tissues of the actual head and compensate for this effect. This is shown by the smaller correlation between the Efield induced on the cortex and the thickness of the tissues surrounding the brain (right) w.r.t the default montage (left). |
References
1 Schork N, Nature 2015; 520 (609–611)
2 Goldberger JJ, JAMA 2013; 309 (2559–2560)
3 Nitsche MA, Pauli W, J. Phys, 2000; 527
4 Bestmann, S. Brain Stim. 2017 10 (865-866)
5 Dmochowski JP, J Neural Eng. 2011 8
6 Ruffini G, Neuroimage 2014; 89 (216-225)
7 Fischer DB, Neuroimage, 2017, 157 (34-44)
8 Miranda PC, Current Opinion in Biomed. Engin 2018 8 (20-27)
9 Wexler, J Medica Ethics, 2015 0 (1-5)
10 https://thebraindriver.com/pages/tdcs-placement-montage-maps-studies
12 Bikson M, Brain Stim 2018; 11(465-480)Bikson M, Brain Stim 2018; 11(465-480)
13 Wurzman R, Annals of Neurology, 2016; 80 (1)
14 Datta A, Front Psychiatry 2012; 3(91)
15 Laakso Brain Stimul 2015; 8 (906-913)
16 Krause B, Front Syst Neurosci 2014; 8(25)




Dear Mr/Mrs,
I need to know more details about transcranial direct current stimulation device …. price, models,…etc.
Kind regards
Greetings Orabi, for information about our products, please contact info@neuroelectrics.com and we’ll help you with your questions.
Thank you.